Normalized body weight was identified as covariate related to enalapril volume of distribution. Transit compartments added using Erlang distribution method to predicted enalapril absorption and enalaprilat formation phases. First-order conditional estimation with interaction was used for parameter estimation.
#Expected objective function nonmem full#
A simultaneous semi-mechanistic population-pharmacokinetic model was developed using NONMEM software, which predicted full profile serum and urine concentrations of enalapril and enalaprilat.
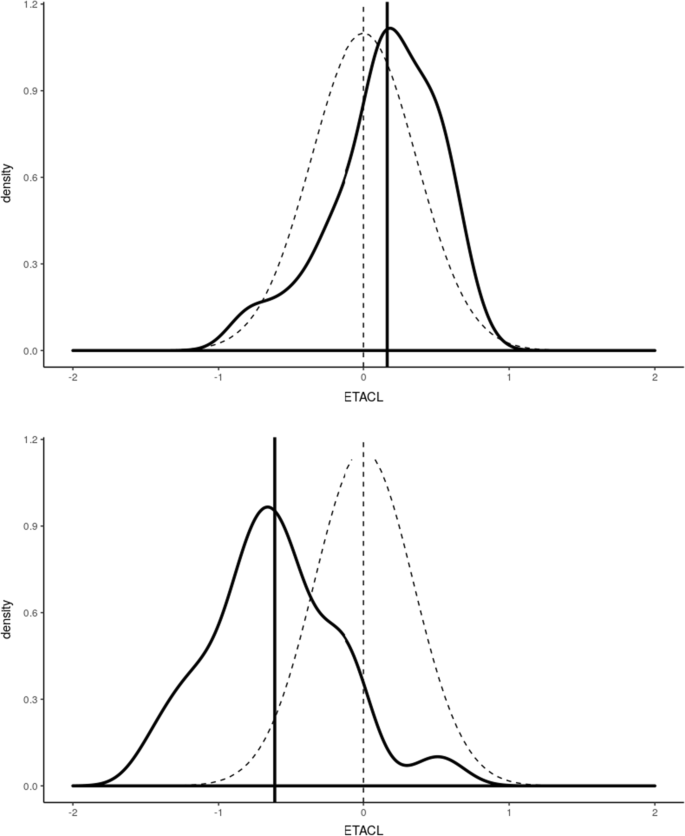

Further, a model informed dosage form population-pharmacokinetic analysis was conducted to evaluate differences in pharmacokinetics of enalapril and its active metabolite enalaprilat when prodrug was administered to 24 healthy adults in a crossover, two periods, two treatments, phase I clinical trial using child-appropriate orodispersible mini-tablets (ODMT) and reference (Renitec ®) dosage formulation. The present analysis was conducted to perform a detailed model informed population pharmacokinetic analysis of prodrug enalapril and its active metabolite enalaprilat in serum and urine.

Burckhardt 1, Jan de Hoon 2, Stephanie Laer 1 and LENA Consortium Muhammad Faisal 1 *, Willi Cawello 1, Bjoern B.
